Developmental Biology Research Group
The developmental biology research group was founded in 2009 at the Department of Genetics of the Biology Insitute of the University of Szeged. The scientific goal of our research group is to gain a better understanding of general cell-biological processes during the formation of gametes, through the examination of specialized cellular organelles of the spermatids. In our laboratory, we use one of the most established model organisms, Drosophila melanogaster (fruit fly), in which a wide range of classic genetic and molecular biology tools can be used to answer scientific questions. Our research group investigate the cellular changes that take place in the developing spermatids. Since the structure of spermatozoa shows a high degree of conservation in the living world, their development is similar in many points to the case of different groups of animals, therefore our results also could contribute to a better understanding of human spermatogenesis.
Our research interest:
Gene expression studies
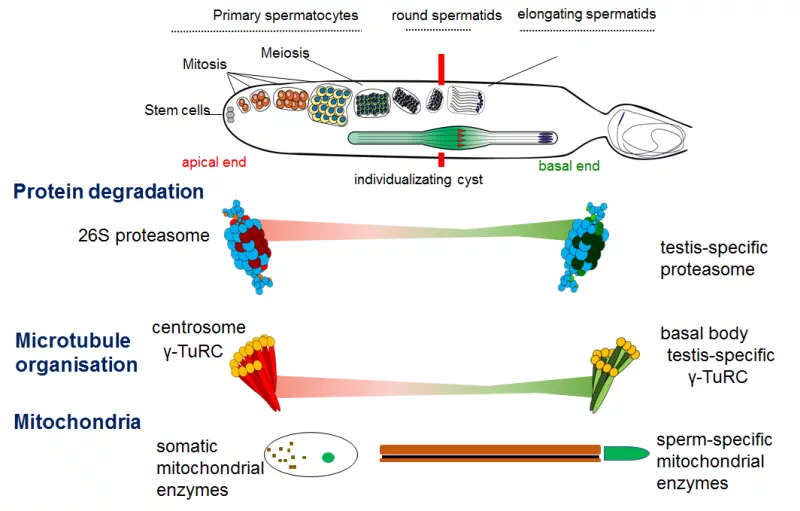
Among animals, it is typical that most genes are expressed in the testis. A large number of testis-specific genes also identified. The number of these varies from species to species, but thousands of these genes are also present in the genome of the fruit fly. Some of them were created by gene duplication. The testis-specific gene expression of the resulting genes enables one of these duplicates to acquire a new function or to specialize in the given organ. Testis-specific gene products are essential for sperm formation. Despite this, the regulation of gene expression in the testis and the appearance of protein products during spermatogenesis are not yet fully understood.
Analysis of Drosophila melanogaster testis transcriptome
Microtubule organizing centers
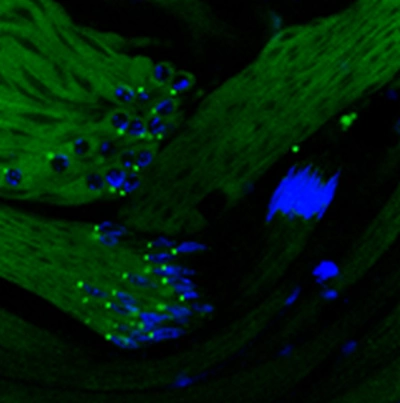
The cytoskeletal elements also have numerous testis-specific paralogs. This is not surprising since the sperm of the fruit fly reaches 1.8mm in length, this transition from round cells requires multiple reorganizations of cellular structures, where cytoskeletal elements play acrutial role. The elements of the gamma-tubulin ring complex (γ-TURC) also have testis-specific paralogs. These genes are required for proper spermatid development and proper basal body formation. The members of the γ-TURC also show mitochondrial association in elongated spermatids, where they might contribute to an alternative microtubule organising centre. Our group aims to clarify the role of the testis-specific γ-TURC members in the basal body and mitochondrial surface.
Microtubule Organizing Centers Contain Testis-Specific γ-TuRC Proteins in Spermatids of Drosophila
Mitochondria and metabolism
The mitochondrion is one of the most complex cell organelle; it is a double membrane bound structure with its own DNA, it gives place to oxidative phosphorilation. The energy producing function of mitochondria is widely known, however there are many other function is associated to the mitochondria, for example it gives structural support for the very long Drosophila sperms. During the differentiation of spermatids, the mitochondria fuse and form two mitochondrial derivatives, which run along the tail part of the sperm. There are many testis-specific mitochondrial genes as well, our group is focusing on genes related to the central metabolic pathways.
Testis-Specific Bb8 Is Essential in the Development of Spermatid Mitochondria
For Students
The topics mentioned above can be used as guidelines for our group's current work. Depending on the topic we use various tools from classical genetics to advanced molecular biological methods and fluorescent microscopy.
Publications
Nádasdi, Bernadett ; Vedelek, Viktor ; Bereczki, Kristóf ; Bukva, Mátyás ; Kozinszky, Zoltan ; Sinka, Rita ; Zádori, János ; Vágvölgyi, Anna (2025). The Influence of Pre-IVF Day 2 TSH Levels on Treatment Success and Obstetric Outcomes: A Retrospective Single-Center Analysis with Machine Learning-Based Data Evaluation. JOURNAL OF CLINICAL MEDICINE Read it
Vedelek, Viktor ; Juma Ochieng, Peter ; Vágvölgyi, Anna ; Nagy, Olga ; Zádori, János ; Sinka, Rita (2025). Analysis of transcript expression and core promoter DNA sequences of brain, adipose tissues and testis in human and fruit fly. INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES Read it
Alzyoud, Elham ; Németh, Dóra ; Vedelek, Viktor ; Szögi, Titanilla ; Tóth, Viktória Petra ; Krecsmarik, Mónika ; Ábrahám, Edit ; Lipinszki, Zoltán ; Sinka, Rita (2024). Versatile gamma-tubulin complexes contribute to the dynamic organization of MTOCs during Drosophila spermatogenesis. COMMUNICATIONS BIOLOGY Read it
Kozinszky, Zoltan ; Bereczki, Kristóf ; Vedelek, Viktor ; Bicskei, Petra ; Tabi, Mariann ; Ekes, Csaba ; Lajkó, Noémi ; Nagy, Olga ; Sinka, Rita ; Vágvölgyi, Anna et al. (2024). Pre- and Procedural Factors Influencing the Success of In Vitro Fertilization: Evaluating Embryo Quality and Clinical Pregnancy in Cases of Tubal Factor Infertility. JOURNAL OF CLINICAL MEDICINE Read it
Vedelek, Viktor ; Jankovics, Ferenc ; Zádori, János ; Sinka, Rita (2024). Mitochondrial differentiation during Spermatogenesis: Lessons from Drosophila melanogaster. INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES Read it
Vedelek, Viktor ; Bicskei, Petra ; Tábi, Mariann ; Bíróné Lajkó, Noémi ; Ékes, Csaba ; Bereczki, Kristóf ; Meixner-Csáti, Zsófia ; Sinka, Rita ; Vágvölgyi, Anna ✉ ; Zádori, János (2024). Endometrium development patterns and BMI groups among in vitro fertilization patients; prognostic aspects. FRONTIERS IN ENDOCRINOLOGY Read it
Datki, Zsolt ; Darula, Zsuzsanna ; Vedelek, Viktor ; Hunyadi-Gulyas, Eva ; Dingmann, Brian J. ; Vedelek, Balazs ; Kalman, Janos ; Urban, Peter ; Gyenesei, Attila ; Galik-Olah, Zita et al. (2023). Biofilm formation initiating rotifer-specific biopolymer and its predicted components. INTERNATIONAL JOURNAL OF BIOLOGICAL MACROMOLECULES Read it
Vedelek, Viktor ; Vedelek, Balázs ; Lőrincz, Péter ; Juhász, Gábor ; Sinka, Rita (2023). A comparative analysis of fruit fly and human glutamate dehydrogenases in Drosophila melanogaster sperm development. FRONTIERS IN CELL AND DEVELOPMENTAL BIOLOGY Read it
Alzyoud, Elham ; Vedelek, Viktor ; Réthi-Nagy, Zsuzsánna ; Lipinszki, Zoltán ; Sinka, Rita (2021). Microtubule Organizing Centers Contain Testis-Specific γ-TuRC Proteins in Spermatids of Drosophila. FRONTIERS IN CELL AND DEVELOPMENTAL BIOLOGY Read it
Bajusz, Csaba ; Kristó, Ildikó ; Abonyi, Csilla ; Venit, Tomáš ; Vedelek, Viktor ; Lukácsovich, Tamás ; Farkas, Attila ; Borkúti, Péter ; Kovács, Zoltán ; Bajusz, Izabella et al. (2021). The nuclear activity of the actin-binding Moesin protein is necessary for gene expression in Drosophila. FEBS JOURNAL Read it
Csonka, Katalin ; Tasi, Zsolt ; Vedelek, Viktor ; Vágvölgyi, Csaba ; Sinka, Rita ; Gácser, Attila (2021). Deciphering of Candida parapsilosis induced immune response in Drosophila melanogaster. VIRULENCE Read it
Jipa, András ; Vedelek, Viktor ; Merényi, Zsolt ; Ürmösi, Adél ; Takáts, Szabolcs ; Kovács, Attila L. ; Horváth, Gábor V. ; Sinka, Rita ; Juhász, Gábor (2021). Analysis of Drosophila Atg8 proteins reveals multiple lipidation-independent roles. AUTOPHAGY Read it
Vedelek, Viktor ; Kovács, Attila L. ; Juhász, Gábor ; Alzyoud, Elham ; Sinka, Rita (2021). The tumor suppressor archipelago E3 ligase is required for spermatid differentiation in Drosophila testis. SCIENTIFIC REPORTS Read it
Vedelek, Balázs ; Maddali, Asha Kiran ; Davenova, Nurgul ; Vedelek, Viktor ; Boros, Imre M. (2020). TERT promoter alterations could provide a solution for Peto’s paradox in rodents. SCIENTIFIC REPORTS Read it
Laurinyecz, Barbara ; Vedelek, Viktor ; Kovács, Attila L ; Szilasi, Kinga ; Lipinszki, Zoltán ; Slezák, Csilla ; Darula, Zsuzsanna ; Juhász, Gábor ; Sinka, Rita (2019). Sperm-Leucylaminopeptidases are required for male fertility as structural components of mitochondrial paracrystalline material in Drosophila melanogaster sperm. PLOS GENETICS Read it
Viktor, Vedelek ; László, Bodai ; Gábor, Grézal ; Bence, Kovács ; Imre, M. Boros ; Barbara, Laurinyecz ; Rita, Sinka (2018). Analysis of Drosophila melanogaster testis transcriptome. BMC GENOMICS Read it
Laurinyecz, Barbara ; Péter, Mária ; Vedelek, Viktor ; L Kovács, Attila ; Juhász, Gábor ; Maróy, Péter ; Vígh, László ; Balogh, Gábor ; Sinka, Rita (2016). Reduced expression of CDP-DAG synthase changes lipid composition and leads to male sterility in Drosophila. OPEN BIOLOGY Read it
Vedelek, V ; Laurinyecz, B ; Kovacs, AL ; Juhasz, G ; Sinka, R (2016). Testis-Specific Bb8 Is Essential in the Development of Spermatid Mitochondria.. PLOS ONE Read it
Group members: